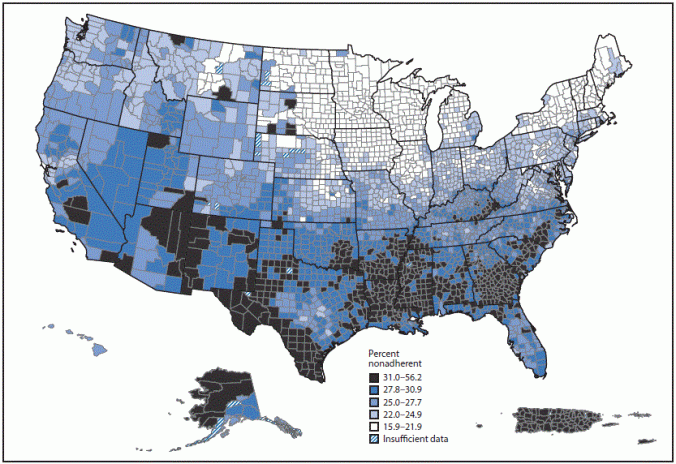
Testosterone and other AAS, which have a schedule III classification by the Controlled Substances Act, may be abused by adults and adolescents, including athletes and body builders.
“Abuse of testosterone, usually at doses higher than those typically prescribed and usually in conjunction with other AAS, is associated with serious safety risks affecting the heart, brain, liver, mental health, and endocrine system,” the FDA notes.
Reported serious adverse outcomes include myocardial infarction, heart failure, stroke, depression, hostility, aggression, liver toxicity, and male infertility. People abusing high doses of testosterone have also reported withdrawal symptoms, such as depression, fatigue, irritability, loss of appetite, decreased libido, and insomnia, the agency says.
Source: FDA Adds New Warnings to All Testosterone Product Labels
It is interesting you no longer see those Low-T commercials on television any more. Really? Just another clever marketing campaign to create a disease that doesn’t exist along with a convenient drug based solution. What side effects?
Read this book. Now.
Source: Bill Moyers Journal . Profile . Melody Petersen | PBS
You must be logged in to post a comment.